When you should call the doctor
- If this is your child’s first blue spell, tell your child’s cardiologist (heart specialist).
- If the frequency of your child’s blue spells increases, tell your child’s cardiologist.
- If the blue spell lasts longer than one minute, take your child to the nearest emergency department.
If the blue spell lasts longer than one minute and there is also a change in your child’s level of alertness or if your child faints, call 911 0r 999
Key points
- During a blue spell, your child may suddenly feel uncomfortable; be cranky or less alert; breathe faster and deeper; turn blue, especially around the mouth and face; faint; or feel faint.
- If your child has a blue spell, stay calm.
- If your child is a baby, place them on their back and bring their knees up to touch their chest. Comfort and soothe them.
- If your child is older, place them on their side and bring their knees up to their chest.
- If the blue spell is long and your child is less alert, or if your child faints, call 911 or 999
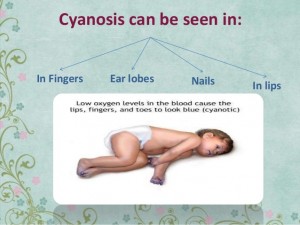
Cyanosis
Cyanosis is a purplish-blue colour to the skin. Newborns with cyanosis are sometimes called “blue babies.”
Cyanosis means that there is less oxygen being carried by the body’s blood cells. In this situation, there may not be enough oxygen being carried to the tissues in the body.
The colour is caused by high levels of reduced (deoxygenated) haemoglobin in the blood near the surface of the skin. Haemoglobin is the molecule that allows red blood cells to carry oxygen. Oxygenated haemoglobin is bright red, while deoxygenated haemoglobin is bluish.
There are two main categories of cyanosis: central and peripheral. Central cyanosis causes a bluish colour in the lips, tongue, and skin, especially the fingers and toes. Peripheral cyanosis is seen only in the fingers and toes.
Cyanosis caused by congenital heart conditions is often seen soon after birth, but sometimes it does not appear until a baby is older.
What causes cyanosis?
Cyanosis may mean that the lungs are not oxygenating the blood properly, or that not enough oxygenated blood is getting to the body.
Central cyanosis has many possible causes, including:
- breathing and lung problems
- heart defects that allow oxygen-poor blood from the right side of the heart to mix with oxygen-rich blood on the left side of the heart, so the blood that is pumped out to the body is low in oxygen; these defects are known as right-to-left shunts
- problems with blood flow to and/or from the lungs: this may be due to abnormal connections of the blood vessels between the heart and lungs, or obstruction of the blood vessels
- fluid build-up in the lungs (pulmonary edema) caused by congestive heart failure
- disorders of hemoglobin, which mean the blood cannot carry oxygen properly
Peripheral cyanosis can also be caused by cold temperature, crying in a newborn, seizures, or shock, among other things.
Congenital heart defects that cause cyanosis include transposition of the great arteries, pulmonary atresia, tetralogy of Fallot, tricuspid atresia, hypoplastic left heart syndrome, truncus arteriosus, and interrupted aortic arch.
What tests will your child need?
Your child may need a variety of tests, including some of the following:
- blood work to measure arterial blood gases, pH, haemoglobin, and blood sugar
- oxygen saturation monitoring
- a chest X-ray
- an electrocardiogram (ECG)
- an echocardiogram
- cardiac catheterization
What is the difference between a sign and a symptom?
Signs and symptoms are not the same things. A sign is a clinical feature of a disease or condition that the doctor looks for. It is something that can be seen or measured. This includes things like redness or the heart murmur, things that the patient may not necessarily feel.
A symptom is something the patient feels or complains about, like fatigue or pain. Symptoms cannot be directly observed. Both signs and symptoms are assessed in order to make a diagnosis.
What influences how common or severe a symptom is?
Symptoms and signs vary according to the type and severity of the heart condition, the child’s activity level, and whether or not the child has been treated for the heart condition.
What are possible symptoms of a heart condition?
- rapid heartbeat
- difficulty breathing (shortness of breath)
- cyanosis (“blue spells”)
- difficulty with feeding (prolonged or ineffective)
- failure to thrive (poor growth)
- decreased exercise tolerance
- palpitations
- fainting/dizziness
- chest pain
What are possible signs of a heart condition?