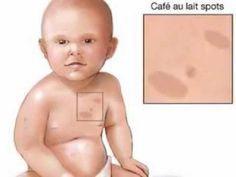
Over the course of the last decade, the definitions of health and disease have been transformed by advances in genetics. Genetic testing has enabled researchers and clinicians to detect inherited traits, diagnose heritable conditions, determine and quantify the likelihood that a heritable disease will develop, and identify genetic susceptibility to familial disorders. Many of the strides made in genetic diagnostics are direct results of the Human Genome Project, an international thirteen-year effort begun in 1990 by the U.S. Department of Energy and the National Institutes of Health, which mapped and sequenced the human genome in its entirety. The increasing availability of genetic testing has been one of the most immediate applications of this groundbreaking research.
A genetic test is an analysis of human deoxyribonucleic acid (DNA), ribonucleic acid (RNA), chromosomes, and proteins to detect heritable disease-related genotypes, mutations, phenotypes, or karyotypes (standard pictures of the chromosomes in a cell) for the purposes of diagnosis, treatment, and another clinical decision making. Most genetic testing is performed by drawing a blood sample and extracting DNA from white blood cells. Genetic tests may detect mutations at the chromosomal level, such as additional, absent, or rearranged chromosomal material, or even subtle abnormalities such as a substitution in one of the bases that make up the DNA. There is a broad range of techniques that can be used for genetic testing. Genetic tests have diverse purposes, including screening for and diagnosis of genetic disease in newborns, children, and adults; the identification of future health risks; the prediction of drug responses; and the assessment of risks to future children.
There is a difference between genetic tests performed to screen for disease and testing conducted to establish a diagnosis. Diagnostic tests are intended to definitively determine whether a patient has a particular problem. They are generally complex tests and commonly require sophisticated analysis and interpretation. They may be expensive and are generally performed only on people believed to be at risk, such as patients who already have symptoms of a specific disease.
In contrast, screening is performed on healthy, asymptomatic (showing no symptoms of disease) people and often to the entire relevant population. A good screening test is relatively inexpensive, easy to use and interpret, and helps identify which individuals in the population are at higher risk of developing a specific disease. By definition, screening tests identify people who need further testing or those who should take special preventive measures or precautions. For example, people who are found to be especially susceptible to genetic conditions with specific environmental triggers are advised to avoid the environmental factors linked to developing the disease.
READ MORE From this below
www.encyclopedia.com/topic/Genetic_Testing.aspx#1
www.encyclopedia.com/topic/Genetic_Testing.aspx#4
What are the types of genetic tests?
ghr.nlm.nih.gov/handbook/testing/uses
Genetic Testing: What You Should Know?
familydoctor.org/familydoctor/en/prevention-wellness/staying-healthy/healthy-living/genetic-testing-what-you-should-know.html
(Go to the bottom of this website below, many many links to this subject very interesting reading)
Explore This Subject
www.nature.com/scitable/topicpage/ethics-of-genetic-testing-medical-insurance-and-651
What does genetic testing involve?
Genetic testing usually involves having a sample of your blood or tissue taken. The sample will contain cells containing your DNA and can be tested to find out whether you are carrying a particular mutation and are at risk of developing a particular genetic condition.
In some cases, genetic testing can be carried out to see if a foetus is likely to be born with a certain genetic condition by testing samples of amniotic fluid (the fluid that surrounds the foetus in the womb) or chorionic villi cells (cells that develop into the placenta) extracted from the mother’s womb using a needle.
Depending on the condition(s) being tested for, the blood or cell samples will then be tested and examined in a genetics laboratory to check for a specific gene, a certain mutation on a specific gene or any mutation on a specific gene.
www.nhs.uk/Conditions/Genetics/Pages/genetic-testing-and-counselling.aspx
How genetic conditions are inherited
Each cell in the body contains 23 pairs of chromosomes. One chromosome from each pair is inherited from your mother and one is inherited from your father.
The chromosomes contain the genes you inherit from your parents. There may be different forms of the same gene – called alleles.
For example, for the gene that determines eye color, you may inherit a brown allele from your mother and a blue allele from your father. In this instance, you will end up with brown eyes because brown is the dominant allele. The different forms of genes are caused by mutations (changes) in the DNA code.
The same is true for medical conditions. There may be a faulty version of a gene that results in a medical condition, and a normal version that may not cause health problems.
www.nhs.uk/Conditions/Genetics/Pages/Facts.aspx
Welcome to UKGTN
The UK Genetic Testing Network is an advisory organization that provides commissioning support to the NHS and DH for NHS patients in the UK CLICK ON THIS LINK www.ukgtn.nhs.uk/