Children with congenital esophageal atresia (EA) and tracheoesophageal fistula (TEF) can have chronic respiratory symptoms
Also with Tof-Oa as Both are the Same Condition But known differently across the world.
Also with Tof-Oa as Both are the Same Condition But known differently across the world.
Respiratory problems in children with esophageal atresia and tracheoesophageal fistula
Published: 5 September 2017
This is very long, and I found interesting, so if you have the time id click on the link below and read on.
There are about 3 Studies, Graphs, and much data
Abstract
Background
Children with congenital esophageal atresia (EA) and tracheoesophageal fistula (TEF) have chronic respiratory symptoms including recurrent pneumonia, wheezing and persistent cough. The aim of this study is to describe the clinical findings of a large group of children with EA and TEF surgically corrected and the instrumental investigation to which they have undergone in order to better understand the patient’s needs and harmonize the care.
Methods
A retrospective data collection was performed on 105 children with EA and TEF followed at Department of Pediatric Medicine of Bambino Gesù Children’s Hospital (Rome, Italy) between 2010 and 2015.
Results
69/105 (66%) children reported lower respiratory symptoms with a mean age onset of 2.2 ± 2.5 years and only 63/69 (91%) performed a specialist assessment at Respiratory Unit. Recurrent pneumonia (33%) and wheezing (31%) were the most reported symptoms. The first respiratory evaluation was performed after surgical correction of gastroesophageal reflux (GER) at mean age of 3.9 ± 4.2 years. Twenty-nine patients have undergone to chest CT with contrast enhancement detecting localized atelectasis (41%), residual tracheal diverticulum (34%), bronchiectasis (31%), tracheal vascular compression (21%), tracheomalacia (17%) and esophageal diverticulum (14%). Fifty-three patients have undergone to airways endoscopy detecting tracheomalacia (66%), residual tracheal diverticulum (26%), recurrent tracheoesophageal fistula (19%) and vocal cord paralysis (11%).
Conclusions
Our study confirms that respiratory symptoms often complicate EA and TEF; their persistence despite medical and surgical treatment of GER means that other etiological hypothesis must be examined and that a complete respiratory diagnostic work-up must be considered.
https://ijponline.biomedcentral.com/articles/10.1186/s13052-017-0396-2
Publish date: November 30, 2016
By: Richard Mark Kirkner Frontline Medical News
Key clinical point: Bioprosthetic materials show progress for reconstruction of large airway defects.
Major finding: Airway defects were successfully closed in all patients, with no postoperative deaths or recurrence of airway defect.
Data source: Eight patients who underwent closure of complex central airway defects with bioprosthetic materials between 2008 and 2015.
Disclosures: Dr Udelsman and coauthors reported having no relevant financial disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Large and complex airway defects that primary repair cannot fully close require alternative surgical approaches and techniques that are far more difficult to perform, but bioprosthetic materials may be an option to repair large tracheal and bronchial defects that have achieved good results, without postoperative death or defect recurrence, in a small cohort of patients at Massachusetts General Hospital, in Boston. Although our results are derived from a limited number of heterogeneous patients, they suggest that closure of noncircumferential large airway defects with bioprosthetic materials is feasible, safe and reliable,” These complex defects typically exceed 5 cm and can involve communication with the esophagus. For repair of smaller defects, surgeons can use a more conventional approach that involves neck flexion, laryngeal release, airway mobilization, and hilar release, but in larger defects, these techniques increase the risk of too much tension on the anastomosis and dehiscence along with airway failure. Large and complex defects occur in patients who have had a previous airway operation or radiation exposure, requiring alternative strategies, the researchers wrote. “Patients in this rare category should be referred to a high-volume center for careful evaluation by a surgeon experienced in complex airway reconstruction before the decision to abandon primary repair is made.
Read the Full report here.
Date: Dec 21, 2015
A microfluidic model of human lung inflammatory disorders provides a new and systematic way to analyse disease mechanisms and test new drug candidates
(BOSTON) — A research team at the Wyss Institute for Biologically Inspired Engineering at Harvard University leveraged its organ-on-a-chip technology to develop a model of the human small airway in which lung inflammatory diseases, such as chronic obstructive pulmonary disease (COPD), the third leading cause of mortality worldwide, and asthma can be studied outside the human body. As reported advanced online on December 21 in Nature Methods, the platform allows researchers to gain new insights into the disease mechanisms, identify novel biomarkers and test new drug candidates.
COPD and asthma are inflammatory reactions in the lung which can be dramatically exacerbated by viral and bacterial infections, as well as smoking. It is known that many of the associated disease processes occur in the conducting airway sections of the lung that shuttle air to and from the alveoli or air sacs. However, much less is known about how inflammation induces distinct pathological processes such as the recruitment of circulating white blood cells and the buildup of mucus, which compromise the lungs of these patients, or how clinical exacerbations are triggered.
“Inspired by our past work using the organ-on-a-chip approach to model the lung alveolus, we created a new microfluidic model of the lung small airway that recapitulates critical features of asthma and COPD with unprecedented fidelity and detail. Now with this micro-engineered human lung small airway, we can study lung inflammatory diseases over several weeks in chips lined by cells from both normal donors and diseased patients to gain better insight into disease mechanisms, as well as screen for new therapeutics,” said Donald Ingber, M.D., Ph.D., the senior author on this work who is leading a multidisciplinary team of Wyss scientists that has been at the forefront of organ-on-chip technology. He is also the Wyss Institute’s Founding Director, the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital, and Professor of Bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
Demand for such opportunities is especially high since small airway inflammation cannot be adequately studied in human patients or animal models and, to date, there are no effective therapies that can stop or reverse the complex and widespread inflammation-driven processes.
“To closely mimic the complex 3D cellular architecture of actual human small airways, we designed a microfluidic device that contains a fully matured human small airway epithelium with different specialized cell types exposed to air in one of its two parallel microchannels. The second channel is lined by a human vascular endothelium in which we flow medium containing white blood cells and nutrients so that the living microsystem can be maintained over weeks. We then modeled inflammatory asthma and COPD conditions by adding an asthma-inducing immune factor or by setting up the system with lung epithelial cells obtained from patients with COPD,” said Remi Villenave, Ph.D., a former postdoctoral fellow in Ingber’s group and the co-first author on the publication. In both cases, the team was not only able to observe highly disease- and cell type-specific changes but could also exacerbate them with agents simulating viral or bacterial infection. CONT….
READ and Watch Video
In the United States, about 500,000 babies, or one in eight, are born prematurely each year. Many of them suffer from apnea of prematurity — a condition in which babies stop breathing for at least 10 seconds as they sleep — and related complications such as hypoxia, which causes insufficient oxygen supply to their tissues. Apnea and hypoxia in premature infants can cause multi-organ damage, developmental delay, and lifelong cognitive deficits, and they represent a major public-health problem. Current apnea and hypoxia therapies, such as caffeine treatment and vigorous manual stimulation, are not always effective. A reliable medical device is badly needed to address these problems.
A multi-disciplinary team of scientists, clinicians and engineers at the University of Massachusetts Medical School, Boston University and the Wyss Institute have developed a versatile, intelligent technology that can inhibit and possibly prevent infant apnea in Neonatal Intensive Care Units (NICUs) and, in the future, for infants at home. The technology consists of an active mattress that senses the cardio-respiratory function of the infant and hardware and software that can help predict when apnea or hypoxia may occur.
The mattress is based on the principle of “stochastic resonance” (SR). This is a counterintuitive phenomenon in which the application of a small amount of “noise” to a complex biological system, such as the human body, increases the sensitivity of that system. In this case, the mattress provides gentle vibration to the baby’s body (well below the baby’s head). This has been shown to promote stable respiration without changing sleep state or waking the infant and can prevent dangerous apneic and hypoxic events from occurring.
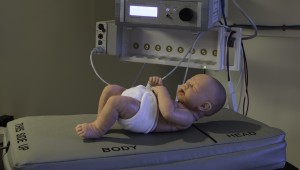
What is fluoroscopy?
 through the part of the body being examined. A continuous X-ray beam is passed through the body part and transmitted to a video monitor so that the body part and its motion can be seen in detail. Fluoroscopy, as an imaging tool, allows doctors to look at many body systems, including the skeletal, digestive, urinary, respiratory, and reproductive systems. Fluoroscopy may be used to evaluate specific areas of the body, including the bones, muscles, and joints, as well as solid organs, such as the heart, lung, or kidneys.
through the part of the body being examined. A continuous X-ray beam is passed through the body part and transmitted to a video monitor so that the body part and its motion can be seen in detail. Fluoroscopy, as an imaging tool, allows doctors to look at many body systems, including the skeletal, digestive, urinary, respiratory, and reproductive systems. Fluoroscopy may be used to evaluate specific areas of the body, including the bones, muscles, and joints, as well as solid organs, such as the heart, lung, or kidneys.Fluoroscopy is used in many types of exams and procedures including:
Barium X-rays
In barium X-rays, fluoroscopy used alone allows the doctor to see the movement of the intestines as the barium moves through them.
Cardiac catheterization
In cardiac catheterization, fluoroscopy is used to help the doctor see the flow of blood through the coronary arteries to check for arterial blockages.
Arthrography
X-ray to view a joint or joints.
Placement of intravenous (IV) catheters (thin, hollow tubes put into veins or arteries)
For IV catheter insertion, fluoroscopy is used to guide the catheter into a specific location inside the body.
Intravenous pyelogram (IVP)
X-ray of the kidneys, bladder, and ureters.
Hysterosalpingogram
X-ray of the uterus and fallopian tubes.
Percutaneous vertebroplasty
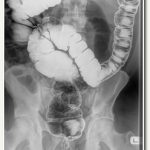 A procedure used to treat compression fractures of the vertebrae (bones) of the spine.
A procedure used to treat compression fractures of the vertebrae (bones) of the spine.
Intravenous pyelogram (IVP)
X-ray of the kidneys, bladder, and ureters.
Hysterosalpingogram
X-ray of the uterus and fallopian tubes.
Percutaneous vertebroplasty
A procedure used to treat compression fractures of the vertebrae (bones) of the spine.
Fluoroscopy is also used for:
Lumbar puncture
Biopsies
Locating foreign bodies
Guided injections into joints or the spine
Fluoroscopy may be used alone or may be used along with other diagnostic procedures. There may be other reasons for your doctor to recommend fluoroscopy.
http://healthlibrary.brighamandwomens.org/library/Encyclopedia/92,P07662#sthash.EMkaGNoN.dpuf
What does a chest x-ray show
Chest X-rays are used to determine the cause of symptoms such as chest pain, trouble breathing, persistent coughing and coughing up blood, reports MedlinePlus. They are also used to diagnose or rule out conditions such as lung disease and tuberculosis. There are many causes of an abnormal chest X-ray. A physician is able to diagnose pneumonia, lung tumours, collapsed the lung, scarring of the lung tissue and other respiratory problems using this test. If the heart is an abnormal size or shape, a physician is able to see the abnormality on a chest film. Chest X-rays also show problems with the position and shape of the large arteries. Because the ribs and spine are visible on a chest film, this type of X-ray is also used to diagnose osteoporosis, rib fractures and spine fractures.
Chest X-rays use a small amount of radiation. In fact, a person receives a similar amount of radiation from a chest X-ray as he would from 10 days of natural radiation exposure.
X-ray machines are designed to permeate the surfaces of lighter objects and materials, such as skin and soft tissues. While beams pass easily through these body parts, they are not absorbed by denser materials, such as bone. The reflection of X-ray beams from dense objects appears as light areas on X-ray films, which identify bone structures and illuminate the skeleton.
This process starts with the generation of electricity, which happens when the machine is turned on. Electrical energy is then carried through a compressed X-ray tube, which transforms energy into multiple X-ray beams. These beams are highly concentrated and exist in various levels of energy. Beams with low energy are blocked, while high-energy beams pass through dense surfaces to provide technicians with a clear image of the skeleton.
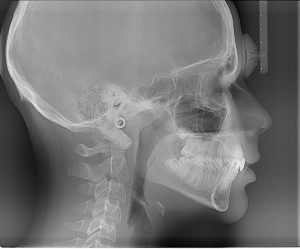
Is ideal if you don’t have a prescription for CPAP or haven’t been diagnosed yet with sleep apnoea. These affordable and discreet, in-home sleep studies can offer a fast-track route to CPAP therapy.
http://www.eu-pap.co.uk/sleep-studies.html?gclid=CM29gcSmwsoCFYMSwwod1hMCGQ
Sleep Studies in Children: What to Expect During Your Child’s Sleep Study
Why Would a Doctor Order a Sleep Study Test?
There are several obvious reasons a child’s doctor may order a sleep study (polysomnogram) for a child: some kids have problems like night terrors, frequent waking, loud snoring, or sleepwalking. There are several less obvious reasons a doctor might order a sleep study: an Ear, Nose, and Throat (ENT) doctor may notice enlarged tonsils and prescribe a sleep study. Persistent bedwetting and some cases of Attention Deficit/Hyperactivity Disorder have been linked to children’s sleep disorders.
Why Our Son’s Sleep Study was Ordered: Our Personal Experience
Our son had constant ear infections and was due to have his third set of ventilation tubes placed (these are also known as grommets, or “ear tubes”). It is standard practice to remove the adenoids when the third set of tubes are placed, as enlarged adenoids can contribute to middle ear fluid. Our ENT wanted to determine if we should remove our son’s tonsils at the same time: to determine if his tonsils were posing any difficulties, she ordered a sleep study. In this case, our son did not present with any obvious sleep disorder (though he had never slept through the night, at two years old this is not an uncommon occurrence). The paediatric sleep study was simply ordered to determine whether he needed a tonsillectomy and adenoidectomy.
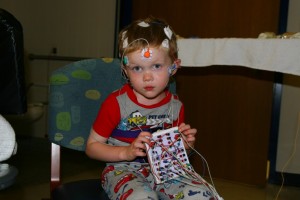
A sleep study test is an overnight procedure. An impressive amount of monitoring equipment will be attached to the child during the sleep study. It is a good idea to prepare your child for a lot of “stickers” and “strings” (the patches and leads that will be attached). Fortunately, no part of a sleep study causes pain: a child can be reassured that there will be no “ouchies” during the test. The most difficult part of a sleep study (for most children) is the placement of a nasal cannula in a child who has never worn one. This is a small tube that runs under the nose, with two small prongs that fit into the nostrils. This is usually placed at the end of the sleep study set-up.
(Watch Viedo’s of a sleep study on the link under this photo)
http://hubpages.com/health/Sleep-Studies-in-Children-What-to-Expect#
CPAP Humidifiers
http://www.eu-pap.co.uk/cpap-sleep-apnoea-products/cpap-machine-humidifiers.html
Sleep Disorders
Episodic apnea leading to asphyxia is a relatively common disorder of young children. Important apnea syndromes include apnea of prematurity, “narrow upper airway syndrome,” congenital hypoventilation syndrome, breath-holding spells, and “near-miss” sudden infant death syndrome. More recently described syndromes include apnea associated with feedings, regurgitation or gastroesophageal reflux and apnea initiated by epileptic seizures. Apnea occurring during wakefulness is common and may be related to that occurring during sleep. Knowledge of the clinical features and pathophysiology of these various kinds of apnea is important in their management.
http://europepmc.org/abstract/med/3906304
What Is Sleep Apnea?
Sleep apnea (AP-ne-ah) is a common disorder in which you have one or more pauses in breathing or shallow breaths while you sleep. Breathing pauses can last from a few seconds to minutes. They may occur 30 times or more an hour. Typically, normal breathing then starts again, sometimes with a loud snort or choking sound.
Sleep apnea usually is a chronic (ongoing) condition that disrupts your sleep. When your breathing pauses or becomes shallow, you’ll often move out of deep sleep and into a light sleep. As a result, the quality of your sleep is poor, which makes you tired during the day. Sleep apnea is a leading cause of excessive daytime sleepiness.
Overview
Sleep apnea often goes undiagnosed. Doctors usually can’t detect the condition during routine office visits. Also, no blood test can help diagnose the condition. Most people who have sleep apnea don’t know they have it because it only occurs during sleep. A family member or bed partner might be the first to notice signs of sleep apnea. The most common type of sleep apnea is obstructive sleep apnea. In this condition, the airway collapses or becomes blocked during sleep. This causes shallow breathing or breathing pauses.
http://www.nhlbi.nih.gov/health/health-topics/topics/sleepapnea
However, CPAP therapy is designed to manage apnea, not to cure it. The machine must be worn throughout each night to provide maximal benefit.
http://www.sleep-apnea-guide.com/cpap-machine.html
Children and C-Pap: Adjusting to Continuous Positive Airway Pressure
Why a Child Would Need C-Pap
What is obstructive sleep apnea? Obstructive sleep apnea is when there is a physical obstruction preventing a breath, or causing a shallow breath (hypopnea) to occur. For the vast majority of children, the cause is enlarged tonsils, which can be surgically removed. In rarer circumstances, children have obstructions caused by floppy airway tissue, neuromuscular weakness, or other physiologic causes which cannot be surgically corrected.
Children with persistent obstructive sleep apnea (and sometimes central sleep apnea) that cannot be surgically corrected may be prescribed a CPAP machine. A CPAP provides a continuous stream of air to splint open the airway while the child is sleeping: this prevents the apneas and hypopneas which cause obstructive sleep apnea. Some children do not tolerate CPAP and must use a bi-level system (BiPAP) or an automatically adjusting airflow system (APAP) for proper breath support and carbon dioxide clearance.
My own son has persistent obstructive sleep apnea, caused by a floppy airway (a condition known as laryngomalacia). Despite surgery, the obstructive apnea could not be helped: he now uses a CPAP machine to help him breathe at night.
The C-Pap Titration: Sleep Study with C-Pap
It is extremely helpful if the parents have access to a CPAP mask prior to the sleep study test. This will help lessen anxiety about the process once the child arrives at the sleep lab/hospital for the sleep study. Let the child play with the mask, and have them wear it while watching TV or playing quiet games. This is called “desensitization” and is an important step in getting a child to accept CPAP therapy.
Once the child’s sleep neurologist or pulmonologist feels that CPAP is a necessary treatment, a CPAP titration will be ordered. This is a sleep study where the CPAP pressure is adjusted to the point where almost all apneas and hypopneas disappear.
The sleep study (polysomnogram) is very similar to ones the child has had prior to the CPAP titration. The technologist will monitor EEG patterns, EKG tracings, leg movement, carbon dioxide levels, oxygen saturation levels, chest wall movements, eye movements, and mouth movements. In addition to the normal polysomnogram equipment, a CPAP mask will be used throughout the study. This will be placed over the nasal cannula which measures respiratory rate and carbon dioxide output.
The technologist will slowly increase the CPAP air pressure until the hypopneas and apneas disappear – the aim is to find the least amount of pressure that will eliminate the obstructive sleep apnea. This information is sent to the sleep neurologist or the pulmonologist, who will then forward the prescription information to a Home Healthcare Company.
In my son’s case, there was no mask available prior to the sleep study. This meant he had to adjust to wearing the mask and the new sensation of blowing air while going through the sleep study test. While he managed to make it through the night and sleep enough to determine the necessary air pressure, it was a difficult night for all involved. Several children’s hospitals have recognized the unique needs of children adjusting to CPAP and provide behavioral therapy prior to the CPAP titration. It is wise to determine if this service is available in a local children’s hospital, as it can make the testing process easier for everyone.
Why is it Important to Treat Pediatric Sleep Apnea?
The vast majority of children will not have sleep apnea or will have sleep apnea which is correctable via a tonsillectomy. Some children will require support with a C-Pap (or BiPAP) machine. Why must sleep apnea in kids be treated? Pediatric sleep apnea has been indicated in a wide variety of childhood conditions.
Bringing the C-Pap Home (Loads more to read on this visit the site)
http://hubpages.com/health/Children-and-C-Pap-Adjusting-to-Continuous-Positive-Airway-Pressure#
Responsive website designed & developed by
![]()